
SOLVED: How much heat is needed to raise the temperature of a 1.0 kg steel pot containing 2.0 kg of water from 25°C to the boiling point? (Lv = 540 kcal/kg, Cwater =

List of the available materials with theirs melting, boiling points and... | Download Scientific Diagram

Selection of stainless steels for handling phosphoric acid (H3PO4) – British Stainless Steel Association
Can a metal melt or boil from extremely low pressure like water or similar substance boils at low temperatures? - Quora
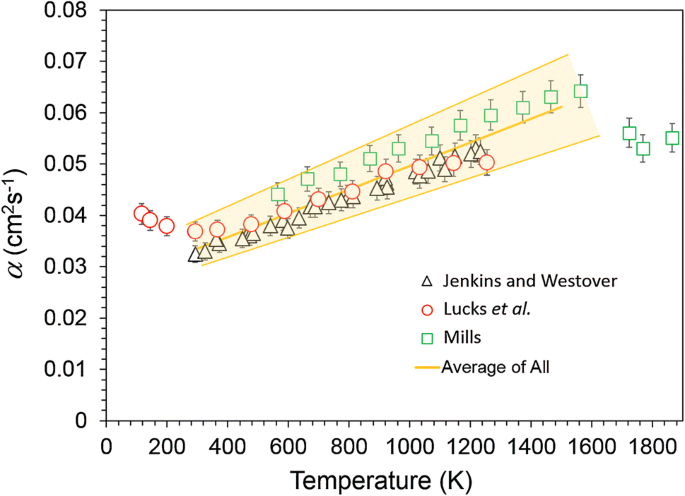


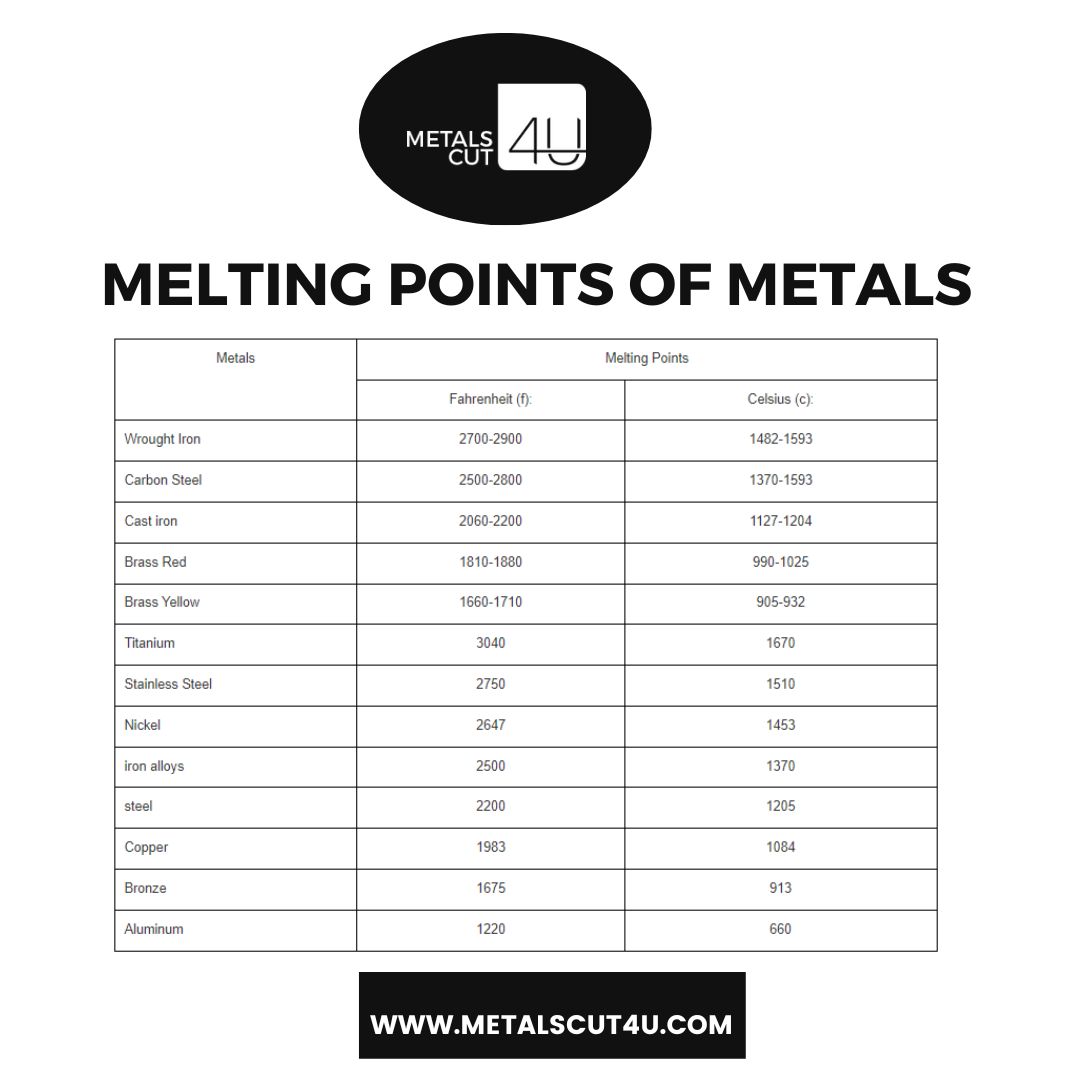




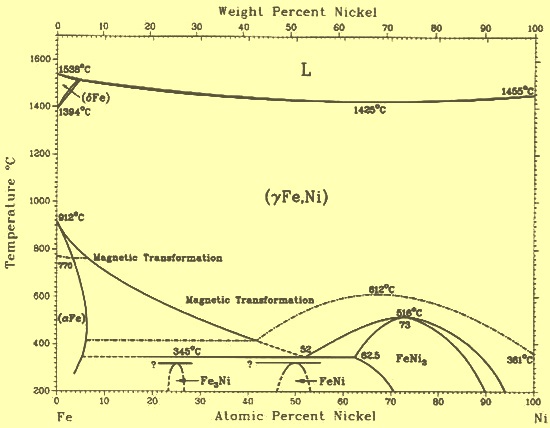
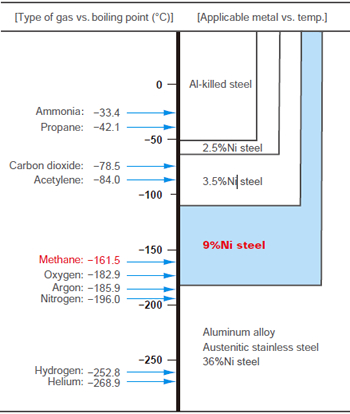





:max_bytes(150000):strip_icc()/sugar-and-water-boiling-in-saucepan-close-up-98359543-5790aebc5f9b584d2005f7e7.jpg)

